Induced Pluripotent Stem Cell: Breakthrough in Medicine
Induced pluripotent stem cell (iPSC) technology has transformed biomedical science by allowing adult somatic cells to be reprogrammed into pluripotent cells capable of becoming virtually any cell type in the body. Since its discovery in 2006, it has provided an ethical and patient-specific alternative to embryonic stem cells, opening doors to personalized disease modeling, drug discovery, and regenerative therapies. As clinical applications expand, it continues to drive innovation in precision medicine and tissue repair.
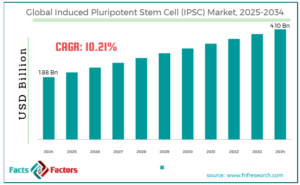
This detailed news article offers a step-by-step exploration of the topic, covering its definition, key insights into technology use, current trends, competitive landscape, consumer preferences, technological innovations, sustainability trends, along with additional perspectives on challenges and the future outlook. With growing investment and clinical progress, iPSC technology is reshaping the future of healthcare.
Definition
An induced pluripotent stem cell (iPSC) is created by reprogramming ordinary adult cells, such as skin or blood cells, through the introduction of specific transcription factors. This reprogramming restores the cells’ ability to self-renew indefinitely and differentiate into all three germ layers, producing neurons, heart cells, liver cells, and many other specialized types.
It avoids the ethical issues of embryonic stem cells while offering the advantage of being derived from a patient’s own tissue, minimizing immune rejection risks. It serves as a powerful tool for creating “disease-in-a-dish” models, high-throughput drug screening, toxicity testing, and developing cell-based therapies for degenerative diseases.
Key Insights
Generating an induced pluripotent stem cell involves delivering reprogramming factors using safer, non-integrating methods such as mRNA, Sendai virus, episomal plasmids, or small-molecule cocktails. Once established, iPSCs are cultured in defined media and directed toward specific lineages using growth factors and 3D organoid techniques.
CRISPR-Cas9 gene editing is frequently combined with iPSC platforms for precise disease modeling or genetic correction. Automated bioreactors and scalable bioprocessing systems support the production of clinical-grade cells under GMP conditions. These technologies ensure high reproducibility, quality control, and the transition from laboratory research to therapeutic applications.
Current Trends
Clinical trials using induced pluripotent stem cell-derived therapies are advancing for conditions like Parkinson’s disease, age-related macular degeneration, heart failure, and spinal cord injuries. Patient-specific iPSC banks and HLA-matched allogeneic lines are expanding to support broader accessibility.
Integration of genome editing with iPSC technology improves disease modeling accuracy. Automated manufacturing platforms and xeno-free culture systems are becoming standard to meet regulatory requirements. The focus is shifting from basic research toward scalable, off-the-shelf cell therapies.
<<<Read More News – https://fnfnewsblog.com/3057/specialty-chemicals-high-value >>>
Competitive Landscape
The field includes biotechnology companies, pharmaceutical giants, and specialized service providers. Prominent players include FUJIFILM Cellular Dynamics, Fate Therapeutics, BlueRock Therapeutics (Bayer), Lonza, Thermo Fisher Scientific, ReproCELL, Ncardia, and Axol Biosciences.
Competition centers on developing clinical-grade iPSC lines, efficient differentiation protocols, scalable manufacturing, and strong intellectual property portfolios. Partnerships between academia, biotech firms, and big pharma accelerate pipeline development, particularly in cell therapy and drug discovery services.
Consumer Preferences
Pharmaceutical companies and researchers demand high-quality, genetically stable iPSC lines with excellent differentiation potential and comprehensive characterization data. Preferences lean toward GMP-compliant, xeno-free systems that support regulatory approval and large-scale production.
Therapy developers seek allogeneic “off-the-shelf” products with low immunogenicity, while academic users value cost-effective research-grade lines. Overall, users prioritize safety, consistency, scalability, and reliable technical support when working with technology.
Technological Innovations Driving Progress
Safer reprogramming methods using non-viral and chemical-only approaches have significantly improved efficiency and reduced genomic risks. Advanced 3D organoid and bioprinting technologies enhance the maturation and functionality of iPSC-derived tissues.
AI platforms now optimize reprogramming protocols and predict differentiation outcomes. Closed-system automated bioreactors enable cost-effective, large-scale manufacturing of clinical-grade cells. Integration of CRISPR editing with iPSC systems allows precise gene correction for therapeutic applications.
Sustainability Trends and Environmental Considerations
The induced pluripotent stem cell field promotes sustainability by reducing dependence on animal-derived materials through xeno-free and chemically defined culture systems. Scalable manufacturing platforms minimize resource consumption compared to traditional animal-based drug testing.
Trends include the adoption of renewable energy in biomanufacturing and efforts to recycle culture consumables. By enabling more efficient drug screening and reducing animal testing, iPSC technology contributes to greener and more ethical biomedical research practices.
Challenges
Potential tumorigenicity from residual undifferentiated cells or genetic instability requires rigorous safety testing and purification steps. Achieving cost-effective, large-scale manufacturing while maintaining consistent quality remains a major technical challenge.
Regulatory pathways for iPSC-derived therapies are complex and differ across regions. Long-term engraftment, functional integration in patients, and immune compatibility in allogeneic settings need further validation. High development costs also limit accessibility for smaller research groups.
Future Outlook
The induced pluripotent stem cell sector is expected to grow substantially as clinical pipelines mature and manufacturing technologies improve. Wider adoption in personalized regenerative therapies, drug discovery, and disease modeling is anticipated in the coming years.
Integration with synthetic biology, AI, and advanced delivery systems will unlock additional applications. With continued emphasis on safety, scalability, and regulatory alignment, the promises are to revolutionize treatment for degenerative diseases and advance precision medicine globally.

![Fire Sprinkler Heads Market [Newest Report] Size Report 2034 Fire Sprinkler Heads Market](https://fnfnewsblog.com/wp-content/uploads/2026/05/Fire-Sprinkler-Heads-Market-150x150.jpg)
